There is an active endogenous nitrogen cycle in humans wherein nitrosating agents that arise from nitrite under acidic gastric conditions react readily with nitrosatable compounds, especially secondary amines and amides, to generate N-nitroso compounds. 
The underlying mechanism for the carcinogenicity determination is endogenous nitrosation that results in the formation of N-nitroso compounds, some of which are known carcinogens. IARC concluded that ingested nitrate under conditions that result in endogenous nitrosation is probably carcinogenic to humans (Group 2A). Meta-analysis of prospective, case-control, and cohort studies reported greater risks for colorectal cancer associated with consumption of processed meat. An increased risk of non-Hodgkin's lymphoma and urinary bladder was reported in some studies but not others at similar exposure levels of nitrate in drinking water. These were reviewed by the International Agency for Research in Cancer (IARC) and included studies of: (1) cancers of the colon, liver, pancreas, and rectum in Canada, China, Slovakia, and Thailand (2) leukemia and lymphoma in Canada, China, Egypt, Finland, Italy, Slovakia, the United Kingdom, and the USA (3) gastric and esophageal tumors in China, Columbia, Costa Rica, Denmark, Japan, Netherlands, Poland, Scotland, Spain, Sweden, the United Kingdom, and the USA (4) tumors of the nervous system, mainly brain, in Australia, Canada, England, France, Germany, Israel, and the USA and (5) genital and urinary tract tumors in Denmark, Egypt, Germany, Slovakia, Spain, and the USA. Various ecological studies, case-control studies, and cohort studies conducted worldwide on the relationship between human exposure to nitrate and the risk for various cancers reported inconsistent results. NTP chronic bioassay studies in rats and male mice did not show carcinogenicity where nitrate was administered alone in drinking water or diet (three studies in mice and four studies in rats) or was coadministered with nitrosatable compounds, and when nitrite was given alone in the diet by gavage or in the drinking water to rats and mice. It has been shown to lower the incidence of tumors in animal experiments, and reduce the risk for cancer that is associated with ingested nitrite in epidemiological studies. Ascorbic acid is an inhibitor of nitrosation reactions. 
Nitrosamines need to be activated metabolically by cytochrome P450 enzymes to electrophilic intermediates to exert a carcinogenic effect, while nitrosamides are direct-acting carcinogens. Nitrosating agents (e.g., nitrous acid and nitrous anhydride) that arise from nitrite under acidic gastric conditions can react with amines or amides to form nitrosamines or nitrosamides, and the induction of tumors in animals via endogenous synthesis of N-nitroso compounds has been demonstrated. Nitrosating agents can react under certain conditions with nitrosatable compounds to form N-nitrosamines and N-nitrosamides, some of which are animal carcinogens. 
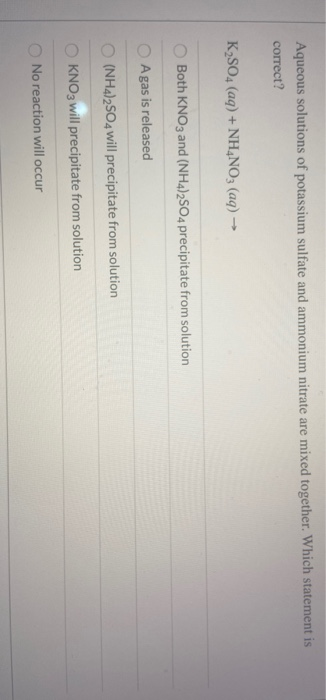
Nitrosating agents can be ingested from food and drinking water, and synthesized from ingested nitrate and nitrite. Sodium and potassium nitrate have been tested for potential carcinogenicity, alone and in combination with nitrosatable compounds. Fan, in Encyclopedia of Toxicology (Third Edition), 2014 Carcinogenicity Empirical Formula Of Precipitate Х 5 ? It will give AgBr light yellow precipitate which solve in conc. The following table summarises which combination will form solids (precipitates) in solution.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
